





Lost your password? Please enter your email address. You will receive a link and will create a new password via email.
Please briefly explain why you feel this question should be reported.
Please briefly explain why you feel this answer should be reported.
Please briefly explain why you feel this user should be reported.
anaphase of mitosis
Anaphase
Anaphase
See lessCombinations (stats and prob)
$(6-2)!=4!$ $4!= 4\times 3 \times 2 \times 1=24$
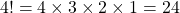
See lessMetabolism and Survival
According to Charles E. Ophardt (2003), The "nitrogen or amino acid pool" is a grand mixture of amino acids available in the cell derived from dietary sources or the degradation of protein. Since proteins and amino acids are not stored in the body, there is a constant turnover of protein. Some proteRead more
According to Charles E. Ophardt (2003), The “nitrogen or amino acid pool” is a grand mixture of amino acids available in the cell derived from dietary sources or the degradation of protein. Since proteins and amino acids are not stored in the body, there is a constant turnover of protein. Some protein is constantly being synthesized while other protein is being degraded. For example, liver and plasma proteins have a half-life of 180 days or more, while enzymes and hormones may be recycled in a matter of minutes or hours.
Each day, some of the amino acids are catabolized producing energy and ammonia. The ammonia is converted to urea and excreted from the body and represents a drain on the nitrogen pool.
A nitrogen balance is achieved by a healthy person when the dietary intake is balanced by the excretion of urea wastes. If nitrogen excretion is greater than the nitrogen content of the diet, the person is said to be in negative nitrogen balance. This is usually interpreted as an indication of tissue destruction. If the nitrogen excretion is less than the content of the diet, a positive nitrogen balance indicates the formation of protein
See lessNewtons question
"Every action has an equal and opposite reaction". This is Isaac Newton's third law. When a rocket is taking off, burning fuel creates a push on the front of the rocket pushing it forward.
“Every action has an equal and opposite reaction”. This is Isaac Newton’s third law. When a rocket is taking off, burning fuel creates a push on the front of the rocket pushing it forward.
See lessSimplify the Equation
$y-(x-5)(x+5)=2y-(x+6)(x+6)$ Open up the brackets $y-(x^{2}-25)=2y-(x^{2}+12x+36)$ $y-x^{2}+25=2y-x^{2}-12x-36$ $x^{2}-x^{2}+12x=2y-y-36-25$ $12x=y-61$ $\therefore 12x-y=-61$
Open up the brackets
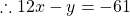
See lessSolve the equation
$\dfrac{4a}{12}+\dfrac{6a}{18}=\dfrac{12a}{24}-\dfrac{16a}{32}$ Divide by the common terms $\dfrac{a}{3}+\dfrac{a}{3}=\dfrac{a}{2}-\dfrac{a}{2}$ $\dfrac{2a}{3}=0$ Cross multiply $2a=0$ $\therefore a=0$
Divide by the common terms
Cross multiply
See lessscience chemistry
1 Kilopascal (kPa) =0.00986923266 atmospheric pressure (atm). To convert kPa to atm, multiply the kPa value by 0.00986923266 or divide by 101.325. Hence $500kpa= 500 \div 101.325=4.9346163atm$
1 Kilopascal (kPa) =0.00986923266 atmospheric pressure (atm).
To convert kPa to atm, multiply the kPa value by 0.00986923266 or divide by 101.325.
Hence
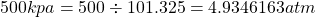
See lessScience chemistry volcanic
Scientists often consider a volcano to be active only if it is showing signs of unrest (i.e. unusual earthquake activity or significant new gas emissions) that mean it is about to erupt.
Scientists often consider a volcano to be active only if it is showing signs of unrest (i.e. unusual earthquake activity or significant new gas emissions) that mean it is about to erupt.
See lessScience History Class
Sir Isaac Newton
Sir Isaac Newton
See lessStatistics and Probability: Binomial Probability Distribution
$2^{4}=16$ The 4 stands for 4 tosses while the 2 stands for 2 outcomes(H,T) Sample space=${HHHH, HHHT,HHTH,HHTT,HTHH,HTHT,HTTH,HTTT,THHH,THHT,THTH,THTT,TTHH,TTHT,TTTH,TTTT}$ Prob.(exactly two heads) = $\frac {6}{16}$
The 4 stands for 4 tosses while the 2 stands for 2 outcomes(H,T)
Sample space=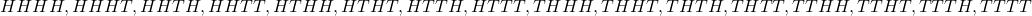
Prob.(exactly two heads) =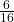
See less